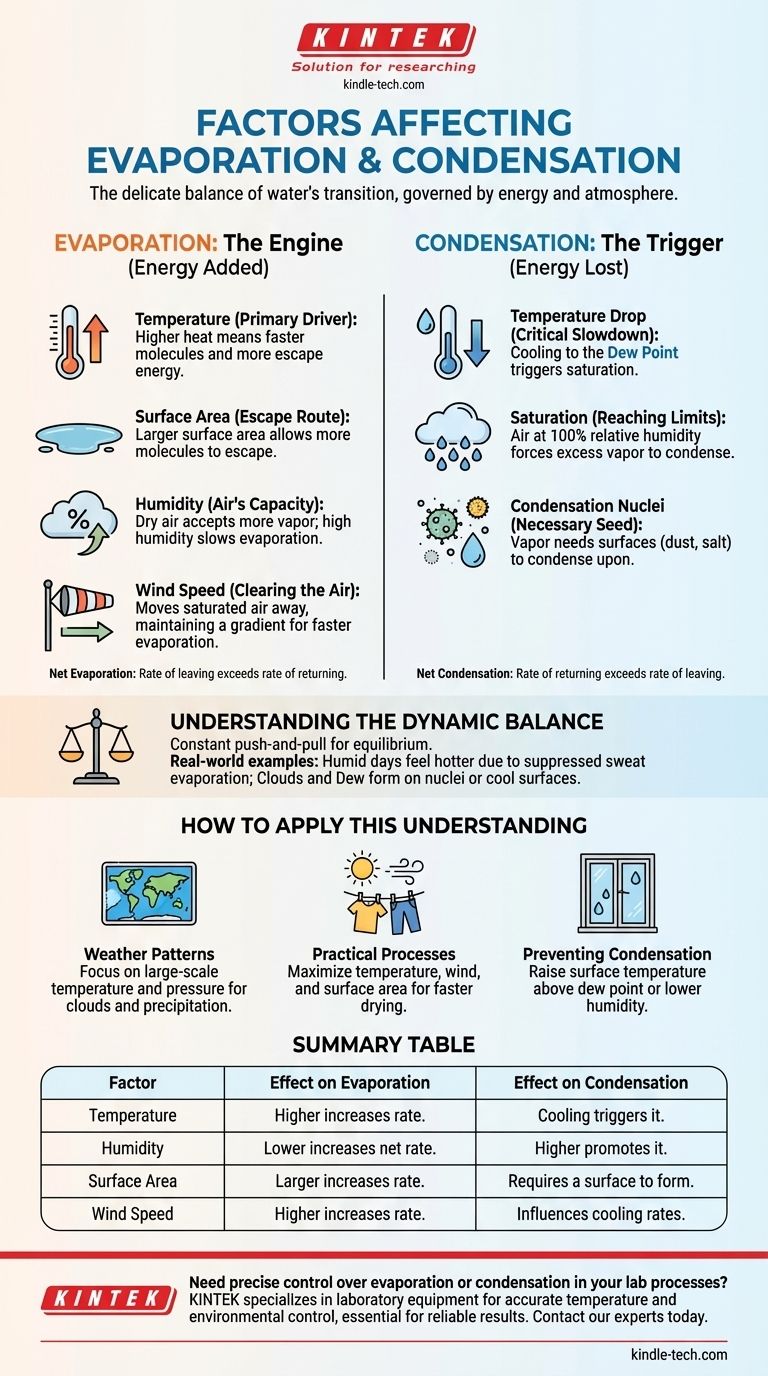
A transição da água entre o estado líquido e gasoso é governada por um delicado equilíbrio de energia e condições atmosféricas. Para a evaporação, os fatores chave são temperatura, área de superfície, umidade e velocidade do vento. Para a condensação, os fatores críticos são uma queda na temperatura até o ponto de orvalho, a quantidade de vapor de água já presente no ar e a presença de superfícies conhecidas como núcleos de condensação.
A evaporação é fundamentalmente um processo impulsionado pela energia que permite à água escapar, enquanto a condensação é a liberação dessa energia à medida que a água retorna ao estado líquido. Compreender essa troca de energia é a chave para entender por que e quando cada processo ocorre.

O Motor da Evaporação: Adicionando Energia e Espaço
A evaporação é o processo onde a água líquida absorve energia suficiente para transicionar para um estado gasoso, ou vapor de água. A taxa na qual isso acontece é controlada por vários fatores interconectados.
Temperatura: O Principal Impulsionador
O calor é uma forma de energia. Quando a temperatura da água aumenta, suas moléculas se movem mais rapidamente e com mais energia.
Essa energia cinética aumentada permite que mais moléculas superem as ligações que as mantêm unidas no estado líquido e escapem da superfície para o ar como vapor.
Área de Superfície: A Rota de Fuga
A evaporação só acontece na superfície de um líquido. Quanto maior a área de superfície exposta ao ar, mais moléculas estão em posição de escapar.
É por isso que uma poça de água espalhada finamente no pavimento evapora muito mais rápido do que a mesma quantidade de água em um copo profundo.
Umidade: A Capacidade do Ar para Água
A umidade relativa mede a quantidade de vapor de água que está atualmente no ar em comparação com a quantidade máxima que ele poderia conter a essa temperatura.
Se o ar já estiver saturado com água (100% de umidade relativa), há pouco "espaço" para mais vapor. Isso retarda significativamente a taxa de evaporação líquida. O ar seco, por outro lado, aceita prontamente mais vapor de água.
Velocidade do Vento: Limpando o Ar
À medida que a água evapora, ela cria uma fina camada de ar úmido diretamente acima da superfície da água. Isso pode retardar a evaporação adicional.
O vento age para afastar essa camada saturada, substituindo-a por ar mais seco. Isso mantém um gradiente de concentração acentuado, incentivando uma taxa de evaporação mais rápida e contínua.
O Gatilho para a Condensação: Perdendo Energia e Encontrando uma Superfície
A condensação é o processo inverso da evaporação. Ocorre quando o vapor de água no ar esfria e se transforma novamente em água líquida, liberando a energia que absorveu durante a evaporação.
Queda de Temperatura: A Desaceleração Crítica
Para que ocorra a condensação, o ar deve esfriar até o seu ponto de orvalho. Esta é a temperatura na qual o ar se torna totalmente saturado com o vapor de água que contém.
À medida que o ar esfria, suas moléculas desaceleram. Uma vez que elas desaceleram o suficiente, as fracas atrações entre elas podem puxá-las de volta para gotículas líquidas.
O Papel da Saturação
A condensação é um resultado direto do ar atingir 100% de umidade relativa. Neste ponto, o ar simplesmente não consegue reter mais água em sua forma gasosa na sua temperatura atual.
Qualquer resfriamento adicional, ou a adição de mais vapor de água, forçará parte desse vapor a condensar em líquido.
Núcleos de Condensação: Uma Semente Necessária
O vapor de água precisa de uma superfície não gasosa para condensar. Na atmosfera, estas são partículas microscópicas de poeira, sal, pólen ou poluentes.
Essas partículas são chamadas de núcleos de condensação, e servem como as "sementes" em torno das quais as gotículas de nuvem se formam. No solo, a condensação se forma como orvalho em superfícies maiores como grama, janelas ou uma lata gelada de refrigerante que esfriaram abaixo do ponto de orvalho.
Compreendendo o Equilíbrio Dinâmico
Evaporação e condensação não são eventos isolados. Estão em uma constante disputa, buscando o equilíbrio. Reconhecer sua interação é crucial para entender fenômenos do mundo real.
O Conceito de Evaporação Líquida
Mesmo quando vemos uma poça encolher, algum vapor de água do ar ainda está condensando de volta na poça.
O que observamos como "evaporação" é na verdade evaporação líquida, o que significa que a taxa de moléculas que deixam o líquido é maior do que a taxa de moléculas que retornam. O inverso é verdadeiro para a condensação líquida.
Por Que Dias Úmidos Parecem Mais Quentes
Nossos corpos esfriam suando. A evaporação desse suor remove o calor da nossa pele.
Em um dia quente e úmido, a alta concentração de vapor de água no ar suprime a taxa de evaporação. Como nosso suor não consegue evaporar eficientemente, perdemos nosso principal mecanismo de resfriamento e sentimos mais calor.
Como Nuvens e Orvalho se Formam
Esses dois fenômenos ilustram perfeitamente os princípios da condensação. As nuvens se formam quando uma parcela de ar sobe, expande e esfria até o seu ponto de orvalho no alto da atmosfera, condensando-se em núcleos suspensos no ar.
O orvalho se forma quando uma superfície no solo, como uma folha de grama, irradia calor e esfria durante a noite para abaixo do ponto de orvalho do ar circundante, fazendo com que o vapor se condense diretamente nela.
Como Aplicar Este Entendimento
Seu objetivo específico determinará quais fatores são mais importantes a considerar.
- Se seu foco principal são os padrões climáticos: Concentre-se nas mudanças de temperatura em larga escala e nos sistemas de pressão do ar, que impulsionam as massas de ar a subir e esfriar, causando condensação (nuvens e precipitação).
- Se seu foco principal é um processo prático (como secar roupa): Você deve maximizar os fatores que aceleram a evaporação — aumentar a temperatura (luz solar), aumentar o vento (uma brisa) e aumentar a área de superfície (espalhar as roupas).
- Se seu foco principal é prevenir condensação indesejada (em janelas ou equipamentos): Você deve aumentar a temperatura da superfície para que ela permaneça acima do ponto de orvalho ou reduzir a quantidade de vapor de água no ar (diminuir a umidade).
Ao compreender esses princípios fundamentais, você ganha a capacidade de prever e até controlar o comportamento da água em inúmeros sistemas naturais e projetados.
Tabela Resumo:
| Fator | Efeito na Evaporação | Efeito na Condensação |
|---|---|---|
| Temperatura | Temperatura mais alta aumenta a taxa. | Resfriamento até o ponto de orvalho desencadeia a condensação. |
| Umidade | Umidade mais baixa aumenta a evaporação líquida. | Umidade/saturação mais alta promove a condensação. |
| Área de Superfície | Área maior aumenta a taxa. | Requer uma superfície (ex: núcleos, grama) para se formar. |
| Velocidade do Vento | Velocidade mais alta aumenta a taxa removendo o ar úmido. | Impacto menos direto, mas pode influenciar as taxas de resfriamento. |
Precisa de controle preciso sobre a evaporação ou condensação em seus processos de laboratório? A KINTEK é especializada em equipamentos e consumíveis de laboratório que fornecem o controle preciso de temperatura e ambiente essencial para resultados confiáveis. Seja você desenvolvendo novos materiais ou realizando análises críticas, nossas soluções o ajudam a dominar essas mudanças de fase fundamentais. Entre em contato com nossos especialistas hoje para encontrar o equipamento certo para suas necessidades específicas de laboratório.
Guia Visual